Performance
Summary of mariPOC® test performance
More detailed information is available in product brochures and user manual.
| Feature | Performance Figure |
|---|---|
| Analysis capacity | High, continuous feed |
| Throughput | 50-100 samples per shift |
| In use stability of test reagents | 4 weeks |
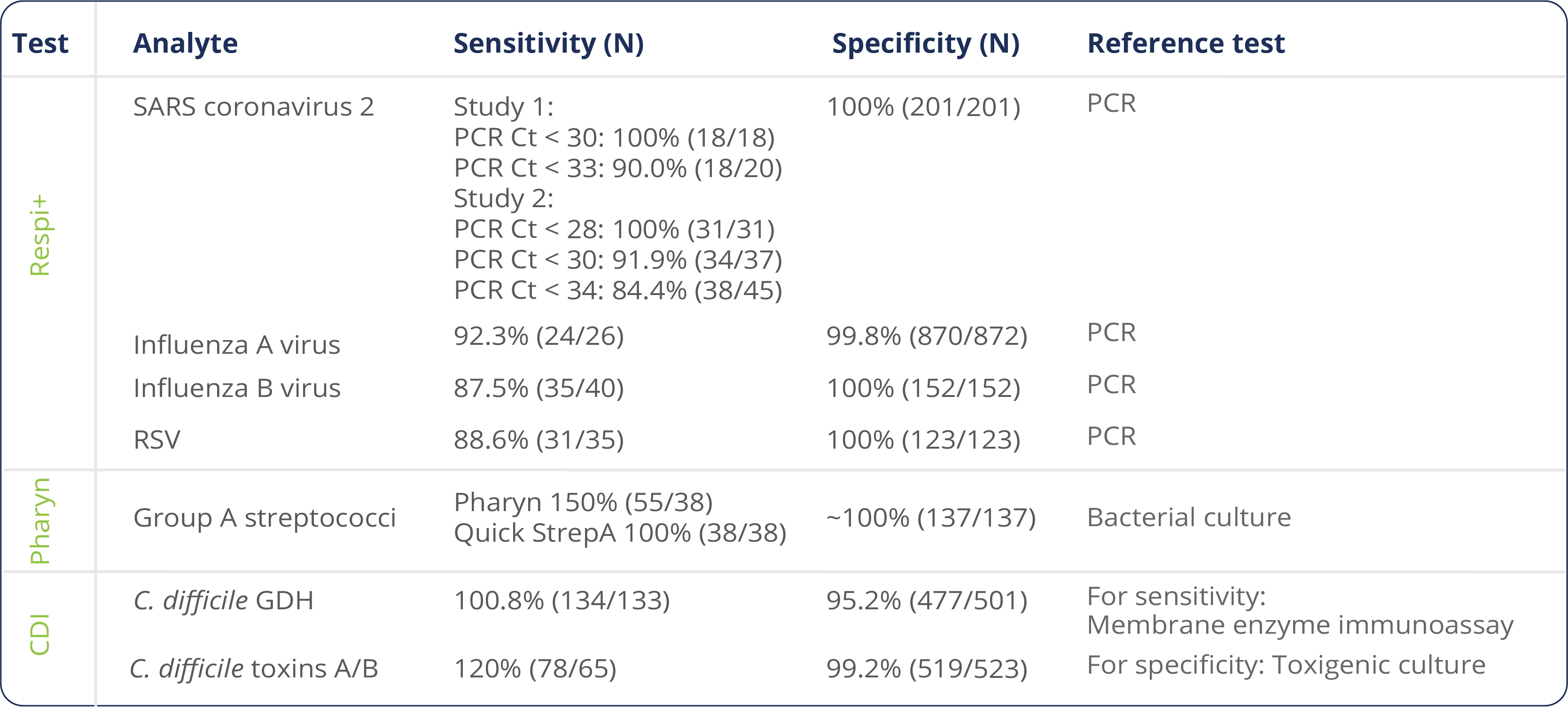
mariPOC®sensitivity and specificity
mariPOC® test system work in continues-feed and random-access mode. New samples can be inserted for automated analyses without waiting for the previous analyses to be finalized. Tens of patient samples can be analysed for multiple pathogens in one plate, and numerous samples can be under analysis simultaneously. The analysis capacity of mariPOC is high. It is exactly what is needed to deliver test results on time also during influenza season when there are lots of samples.
The Limit of detection (LoD) for mariPOC SARS-CoV-2 test method in SARS-CoV-2 and Respi+ tests is *140 TCID50/ml corresponding to 84,500 copies/ml, thus meeting the United Kingdom travel requirement for COVID-19 tests which is > 80% sensitivity at viral loads above 100,000 copies/ml.
mariPOC SARS-CoV-2 test method in Quick Flu+ test meets the United Kingdom travel requirement for COVID-19 tests with 90% sensitivity at 84,500 copies/ml.
*Limit of Detection (LoD) is the lowest concentration of SARS-CoV-2 spiked into nasopharyngeal matrix showing minimum of 95% positivity.
Learn more about our tests performances: